Why Measuring Key Clinical Markers Matters in Cardiogenic Shock
Blog
Cardiogenic shock (CS) remains a critical issue in hospitals nationwide. Despite advances in interventional cardiology, CS is a high-mortality condition, with mortality ranges from 30% to 40%, and 1-year mortality approaches or exceeds 50%.
One of the main challenges is the rapid progression of CS severity. Historically, staging using the Society for Cardiovascular Angiography and Interventions (SCAI) staging system relied heavily on clinical judgement, which often led to inconsistent diagnosis, variable escalation of care, and difficulty comparing outcomes across centers. Kapur et al argued that “the lack of uniform, granular criteria to define hypotension and hypoperfusion may limit its clinical utility.” Standardizing assessment and re-assessing as physiology evolves is therefore critical.
“Criteria for Defining Stages of Cardiogenic Shock Severity” was a large multicenter study by Kapur et al which analyzed over 3,400 CS patients across 17 hospitals across the United States. The purpose of the study was to test parameters that define SCAI stages and explore their utility as predictors of in-hospital mortality in CS.
The authors found that four key parameters: systolic blood pressure (SBP), lactate, alanine transaminase (ALT), and systemic pH were significantly associated with in-hospital mortality and clearly distinguished between SCAI stages.
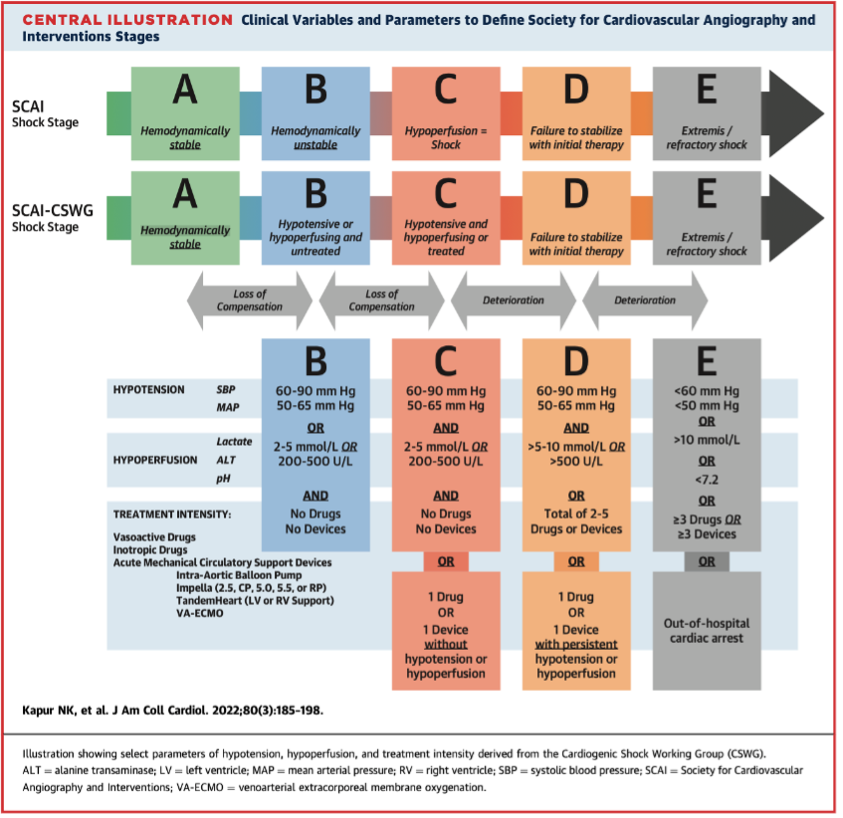
The study also showed that both baseline stage and progression through stages strongly correlated with outcomes, underscoring the importance of tracking these measures early and over time.
Why measuring key parameters together matters
Each parameter – SBP, lactate, ALT and pH reflects a different physiological dimension of CS and when assessed together, give a complete picture of shock severity. This consistency makes SCAI staging more reliable, enabling clinicians to compare patients across hospitals, predict outcomes with greater confidence, and evaluate the impact of interventions.
The insights from Kapur et al. highlight the need for continuous monitoring and dynamic reassessment in CS. This is where the Etiometry platform can help.
How the Etiometry platform can support the SCAI staging system
- Leveraging high-fidelity data to identify and classify cardiogenic shock
- Supports clinician awareness of patient trends by flagging when pre-configured hospital criteria are met.
- Real-time SCAI and other shock staging protocols: Enables teams to continuously monitor patient progression through stages of CS and evaluate the effectiveness of interventions.
- Hospital-wide surveillance: Assists early identification of patient deterioration across the hospital and even from feeder hospitals, not just in the ICU.
- Informational Flags: Identifies patients who meet criteria for patient assessment based on hospital protocols.
- Team-wide coordination: facilitates coordination across the care team, from the front line providers to the consulting physicians.
Contact Us
References
Sinha, S, Morrow, D, Kapur, N. et al. 2025 Concise Clinical Guidance: An ACC Expert Consensus Statement on the Evaluation and Management of Cardiogenic Shock: A Report of the American College of Cardiology Solution Set Oversight Committee. JACC. 2025 Apr, 85 (16) 1618–1641. https://doi.org/10.1016/j.jacc.2025.02.018
Early Prediction of Cardiogenic Shock Using Machine Learning https://pubmed.ncbi.nlm.nih.gov/35911549/
The Lancet: https://www.thelancet.com/journals/lancet/article/PIIS0140-6736%2824%2901818-X/fulltext
References to earlier recognition are based on published research and do not imply predictive or diagnostic functions of the Etiometry Platform.


